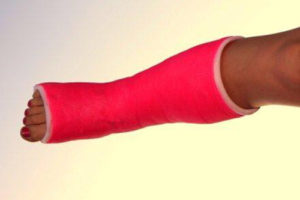
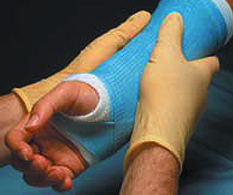
Custom Non-woven Manufacturing Of Non-allergenic Dermal Contact Cast Wrap/medical Padding With Wicking Control
A Medical Company approached Delaware Valley Corporation with a request to develop a dermal (skin) contact product that they hoped would revolutionize the process of making Arm and Leg splints and casts. All their previous development efforts had been unsuccessful. Delaware Valley seemed their last hope.
The theory required a material to be easier and quicker to use than the existing plaster reinforced casting materials that had been in use for many years. The fabric to be developed would permit the creation of splints or casts, for the stabilization of broken bones, in situ on the injured limb, with no need for pre-mixing of materials.
Our task was to create a non-woven fabric that would serve multiple functions. It would be a non-allergenic cushioning material to be placed next to the skin without creating irritation or itching, while functioning as part of the reinforcement media for the cast. The Needle Punch fabric was also to serve as the carrier of the active ingredients of the casting polymer, thus also serving as the outer packaging of the finished composite.
The fabric had to be hydrophobic (water repellant), soft and non-allergenic, compatible with an epoxy based polymer, stretchable within tight deformation parameters, lofty but with specific tensile strengths. One key factor in the development was the ability of the Needle Punched fabric to control the wicking time of water as it was applied to the fabric for activation of the polymer casting material. These required properties had not been achievable using typical woven and knit fabrics.
Delaware Valley met all the requirements after a six month development characterized by applying theory to practice and empirically testing in trial runs. The results speak for themselves as our felt was found to be the only viable candidate for the job, and we continue to produce it 25 years later. It is part of the now universally accepted splinting material used around the world.
Delaware Valley has been able to develop fabrics that solve specific needs and to work hand in hand with potential customers to rapidly solve their requirements for functional fabrics.
Custom Non-allergenic Dermal Contact Cast Wrap/medical Padding With Wicking Control Project Highlights:
| Capabilities Applied/Processes (Needle Punching, Structuring, Die Cutting, Laminating, etc...): | Needle Punching, Precision fiber structuring, Critical fiber selection |
|---|---|
| Tightest Tolerance: | Weight, Tensile Strength, Elongation, Fiber denier |
| Material Type: | Polypropylene |
| Material Thickness: | 0.125 inches |
| Product Length: | 112 yard rolls |
| Product Width: | 54 inches |
| Temperature Range: | Melting point above 320 degrees F (160 deg. C) |
| Color: | White |
| Material Finish (Flat Punch, Velour, Pattern, Coarse, etc...) | Flat punch |
| Resistances (Oil, Water, Grease, etc...): | NA |
| Packaging: | Plastic bagged roll with paper tube core |
| Special Features: | Wicking control, no water solubility, no reactivity to polymer, non-allergenic |
| Secondary Operations Applied: | Cleaned of impurities |
| Industry for Use: | Medical |
| Volume: | 750,000 square yards per year |
| Delivery Time: | Per order |
| Delivery Location: | International |
| Standards Met (Customer Supplied Print, ISO, ASTM, etc...): | ASTM D1777, ASTM D5034, ASTM D5587, ASTM D3776, ISO 9001:2015 |
| Product Name: | Medical pad |
| Project Scope: | See details above |
| Development Work (Prototype, Tensile Test, Porosity Test, Abrasion Test, etc...) | Needling Density versus Wick-ability Testing |
| Drawing Type Accepted: | NA |
| Tooling Cost: | NA |
| Shipping Cost | NA |
| Product Description: | Flat punch, non-woven |
| Product Image File Name: | Cast Wrap |